 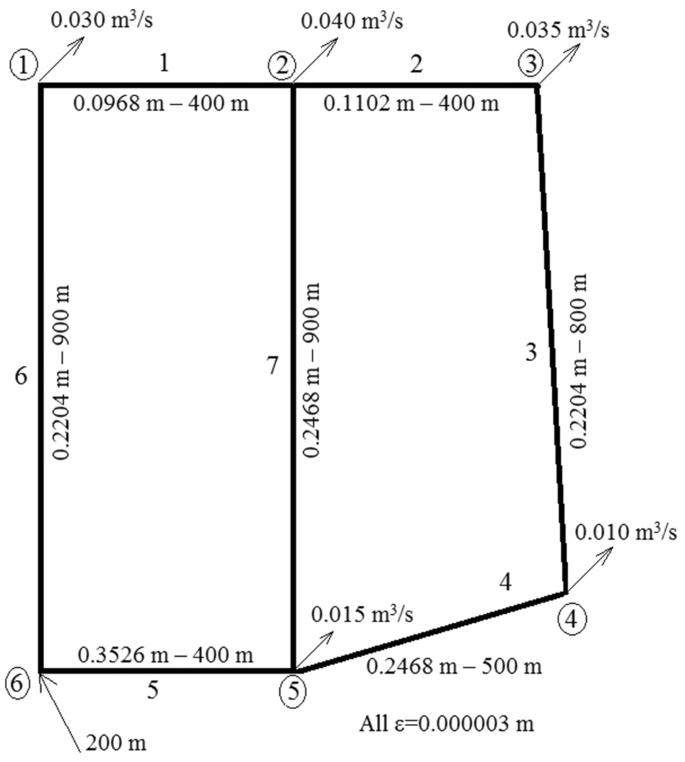
Second, the Ag(I) can migrate across the membrane to the cathode compartment. First, Ag(II) can react directly with water in the anode compartment toįorm oxygen gas (O 2). Three additional reactions that can occur will affect the energy efficiency of the process. 
In combustion processes like the baseline incineration system, these elements yield gases (assuming oxidation is complete), which must be removed in a treatment train, but the treated process gas stream is difficult to analyze prior to release to the atmosphere. This solution can be analyzed and treated further, if necessary, prior to release. In the electrochemical process, the sulfur, phosphorus, and chlorine components of the agent appear in the final effluent as hydrated anions in aqueous solution (sodium is the principal cation). In both processes, carbon is released to the gas-phase primarily as CO 2. The overall reactions are similar to the overall reactions for incineration of VX and HD, but they occur at low temperature (less than 90☌) and close to atmospheric pressure. HD: C 4H 8SCl 2 + 7O 2 + 4NaOH = 4CO 2 + Na 2SO 4 + 2NaCl + 6H 2O In the presence of organics, AgNO 3 + ions oxidize water into intermediates such as hydroxyl radicals that rapidly oxidize the organic species. The Ag(II) species has been shown to exist in the form of AgNO 3 + ions (Po et al., 1968), which impart a brown color to the solution in the absence of organics. When power is applied to the cell, Ag(I) ions are oxidized at the anode to the highly reactive Ag(II). The aggregate volume of all the anode-cathode chambers within a cell is 2.5 m 3.Īt the start of operation, the composition of the anolyte is approximately 8 molar in nitric acid, 0.5 molar in silver nitrate, and 0.02 to 0.03 molar in agent. Thus, the 360-kW power supply unit for a standard module must provide a total of 90 kA and 180 kW to each of the two-cells that make up the module. The anode-cathode chambers are connected in parallel, each pair requiring a normal operating current of 2,000 A at a nominal 2 volts DC. Each FM21 cell comprises 45 anode-cathode compartments, each 10 mm wide by 240 mm high each electrode is separated by a Nafion 1 membrane, which is permeable to cations and water but impermeable to anions ( Figure 5-2). The core reactions take place in two separate 180-kW, electrochemical cells (model ICI FM21), which are connected in parallel through a 360-kW power supply. In addition, laboratory tests conducted at Dounreay since 1987 have demonstrated destruction of 68 organic compounds encountered in industrial wastes, including HD (distilled S-mustard), VX, and GB (another unitary chemical nerve agent).įigure 5-1 is a schematic diagram of the heart of the Silver II process as described by the TPC for destruction of VX and mustard. Most of the TPC's effort to date has been dedicated to operation of a 4-kW pilot plant for destroying inactive fuel solvent composed of 10 percent tributyl phosphate in kerosene. AEA Technology submitted the Silver II technology to the Army for consideration as an alternative technology for agent destruction at the Aberdeen and Newport sites and will therefore be referred to as the TPC (technology proponent company) for the Silver II process in the remainder of this report. It was originally developed in 1987 by AEA Technology at Dounreay, Scotland, as a means for destroying solid and liquid radioactive organic waste streams from the U.K. Silver II is a patented electrochemical process. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
